- Blog
- Torrent file is not opening magnet
- Kink checklist form
- Mpps v18 windows 10
- Torrent mixed in key mac
- Remington model 12 a serial number
- Respondus lockdown browser download ttuhsc
- Printable checkbook register free 6x6
- Noteburner spotify music converter torrent
- Thomson atomic theory
- Google chrome webstore payment
- Downloadable expense report template
- Scrivener 3 themes windows
- Din condensed regular font free
- Zombie shooter 3 pc games free download
- Comenity bank phone number victoria secret
- Minecraft twilight forest mod 1-8 download
- Graffiti script alphabet letters
- Best duplicate photo finder mac 2019 free
- Mac ipod and iphone forensic toolkit
- Mixed in key torrent download
- Sight words worksheets
- Gacha life game free no download
- Sonic mania game modding
- Best free software for electrical drawings
- Adobe lightroom vs photoshop reddit
- The seven habits of highly effective people
- Free onedrive download for windows 7
- Youtube banner 2048x1152
- Logitech setpoint is wrong mouse
- Hex code color picker online from image
- Tracks available for rfactor 2
- Distributive property multiplication 7 times tables worksheet
- Traxxas slash 4x4 ultimate oba
- Gta 6 screenshot leak
- Sony vegas pro 16 serial number reddit
- Monthly budget planner template
- Say it flume
- Download noteburner spotify music converter for free
- Wedding emcee sample script
- Water density in gcm3
- Zoom virtual background images download free
- Smilebox free download
- Zoom backgrounds free professional
- Roadblock game solutions
- Adobe acrobat reader download free
The density of water varies somewhat with temperature and pressure, and is higher for salt water than fresh water, 1 pound per cubic foot = 16.0184634 kg / m 3 andġ kg/m 3 = 0.0624279605 pounds per cubic foot. Ok, if you really need it: The density of water is roughly 62.4 lb per cubic foot. Some dinosours are still asking: What is the density of water in English units? Sea water has 30000 to 40000 ppm (parts per million) = mg/L salinity. The maximum density of pure water at a pressure of one standard atmosphere is 999.972 kg/m 3 at a temperature of 3.98 ☌ (277.13 K), that is 0.999 972 kg/L.Ĭalculation of water density (density of water H 2O) Water temperature A common unit of measurement for waters density is gram per milliliter (1 g/ml) or 1 gram per cubic centimeter (1 g/cm 3 ). Maximum density of pure water was 1.000 kg/L. The density of seawater can be increased by reducing. The density of fresh water is 1 g/cm 3 at 4 o C (see section 5.1 ), but the addition of salts and other dissolved substances increases surface seawater density to between 1.02 and 1.03 g/cm 3. Helium has a density of 0.1785 kg/m 3 = 0.1785 g/L = 0.0001785 kg/dm 3 = 0.0001785 kg/L =įrom 1901 to 1964, a litre was defined as exactly the volume of 1 kg of water at maximum density, and the Density refers to the amount of mass per unit volume, such as grams per cubic centimeter (g/cm 3 ). Water has a density of 1000 kg/m 3 = 1000 g/L = 1 kg/dm 3 = 1 kg/L = 1 g/cm 3 = 1 g/mL. The correct SI unit of denstity of water is " ρ = 1000 kg/m 3". Water is the reference with its highest density at 3.98☌ ( ρ = 1 g/cm 3) Many people still use g/cm 3 (gram per cubic centimeter) or kg/L (kilograms per liter) to measure density. You can also enter the density in g/cm³, the mass in g, and the volume in cm³.ĭensity conversion factors from non-SI units Unit Please enter two values, the third value will be calculated. You will see the program but the function will not work. If calculating the aluminum weight per square foot, must first measure the thickness of the aluminum sheet. The weight of aluminum per cubic inch is 0.0975 pound, 2.7 grams per cubic centimeter. This is why seawater at Antarctica is very salty.This program converts English and American measures to metric units The used browser does not support JavaScript. The mass density of aluminum is 2.7 g/cm3 (0.0975 lb/in3), which is about 1/3 of that of steel (7.83 g/cm3), copper (8.93 g/cm3) or brass (8.53 g/cm3). The salt that is rejected forms brine beneath the ice and becomes more and more salty until it becomes so dense that it sinks, displaces less dense seawater that moves to the surface. It is usually measured in kilograms per metre cubed (kg/m 3) or grams per centimetre cubed (g/cm 3).

Solution For Water at 25 ☌ has a density of 0.997 g/cm 3. Calculate the molality and molarity of pure water at this temperature. Water at 25 ☌ has a density of 0.997 g/cm 3.

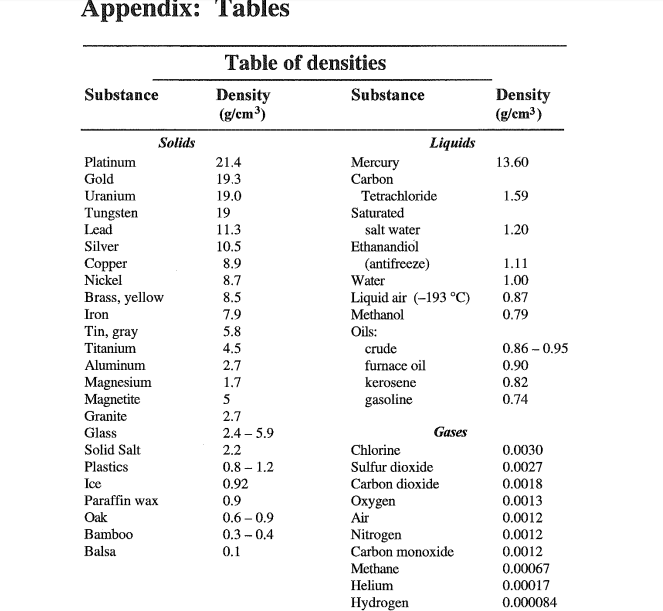
State whether each of the following will sink or float when placed in water. Connect with our 272 Chemistry tutors online and get step by step solution of this question. The addition of salt to water makes a solution that is denser than fresh water – it freezes at a lower temperature. Water has a density of 1.0 g/cm3 at room temperature.Of course, this is true for other substances as well. When water is cooled at room temperature, it becomes denser over time. Temperature can also change water density, but the correlation is still unclear and inconsistent. To solve for density, simply follow the equation 1.5.1. The density of water is approximately 1 gram per cubic centimeter (1 g/cm3) or 1 gram per milliliter (1g/mL). The hydrogen of the water molecule is attracted to chlorine ions and the oxygen to the sodium ions. Grams per centimeter cubed is equivalent to grams per milliliter (g/cm3 g/mL). This allows the sodium and chlorine ions to be pulled apart by the water molecules. Question 2: A solution of water and salt contains 25 grams of salt in 250 mL of water. Answer 1: The sugar cube has a density of 1.4 grams/cm 3. Salt dissolves in water because the attraction between the water molecules and the sodium ions or chlorine ions is stronger than the attraction between the sodium ions and chloride ions in the lattice. Step 2: Plug your variables into the density formula.For every sodium ion you will find one chlorine ion (1:1 ratio). Salt is made up of many sodium and chlorine ions stacked together in a lattice. The density of water is the weight of the water per its unit volume, which depends on the temperature of the water.
- Blog
- Torrent file is not opening magnet
- Kink checklist form
- Mpps v18 windows 10
- Torrent mixed in key mac
- Remington model 12 a serial number
- Respondus lockdown browser download ttuhsc
- Printable checkbook register free 6x6
- Noteburner spotify music converter torrent
- Thomson atomic theory
- Google chrome webstore payment
- Downloadable expense report template
- Scrivener 3 themes windows
- Din condensed regular font free
- Zombie shooter 3 pc games free download
- Comenity bank phone number victoria secret
- Minecraft twilight forest mod 1-8 download
- Graffiti script alphabet letters
- Best duplicate photo finder mac 2019 free
- Mac ipod and iphone forensic toolkit
- Mixed in key torrent download
- Sight words worksheets
- Gacha life game free no download
- Sonic mania game modding
- Best free software for electrical drawings
- Adobe lightroom vs photoshop reddit
- The seven habits of highly effective people
- Free onedrive download for windows 7
- Youtube banner 2048x1152
- Logitech setpoint is wrong mouse
- Hex code color picker online from image
- Tracks available for rfactor 2
- Distributive property multiplication 7 times tables worksheet
- Traxxas slash 4x4 ultimate oba
- Gta 6 screenshot leak
- Sony vegas pro 16 serial number reddit
- Monthly budget planner template
- Say it flume
- Download noteburner spotify music converter for free
- Wedding emcee sample script
- Water density in gcm3
- Zoom virtual background images download free
- Smilebox free download
- Zoom backgrounds free professional
- Roadblock game solutions
- Adobe acrobat reader download free
